FRONTIERS IN MEDICAL CASE REPORTS - Volume 7; Issue 2, (Mar-Apr, 2026)
Pages: 01-08
Print Article
Download XML Download PDF
Adult Bladder Rhabdomyosarcoma Developing Five Years After Fibroepithelial Polyp Removal: A Case Report
Author: Scott Fassas, Nicholas Freidberg, Sam Kwon, Andrew Hall, Michael Whalen
Category: Medical Case Reports
Abstract:
Rhabdomyosarcoma (RMS) is a soft tissue sarcoma that is rarely described in the bladder of an adult patient. We report a case of a bladder rhabdomyosarcoma in a 35-year-old African American male with prior history of benign fibroepithelial polyp removal. Given the unusual presentation of this tumor and the putative etiologic link of malignant transformation from a fibroepithelial polyp, this case report and review of the literature highlight an interesting association that has only once been previously reported. Furthermore, we detail our management of this adult bladder RMS.
Keywords: Rhabdomyosarcoma, Fibroepithelial Polyp, Bladder Tumor, Ileal Neobladder
Full Text:
Introduction
Rhabdomyosarcomas (RMS) are malignant soft tissue tumors that recapitulate the morphologic features and molecular properties of striated skeletal muscle. Although RMS are commonly found in the pediatric population, they are rare in adults, accounting for less than 5% of adult soft tissue tumors (Hawkins et al., 2001). They can arise from any site in the body, most commonly the head and neck, genitourinary organs, extremities, and trunk. In adults, approximately 20% of RMS cases occur in the genitourinary organs (Ahsaini et al., 2018). Due to the rarity of RMS in adults compared to pediatric populations, limited research and lack of standardized protocol may contribute to worse survival outcomes compared to children with similar tumors (Sultan et al., 2009). We report a case of bladder RMS in a 35-year-old African American patient, managed solely with surgical resection.
Case Presentation
A 35-year-old African American male with history of recurrent urinary tract infections, lower urinary tract symptoms, and a benign urinary bladder fibroepithelial polyp (FEP) (status post transurethral resection 5 years prior with no follow-up cystoscopy), presented with intermittent flank pain and new onset hematuria. He reported longstanding urinary frequency, incomplete emptying, and nocturia. He had no relevant family history and social history was significant for tobacco smoking. Physical examination demonstrated moderate left flank tenderness with unremarkable vital signs. Urinalysis showed mild hematuria and proteinuria with negative urine culture. Abdominal/pelvic CT with contrast displayed an irregular 5x4cm filling defect in the bladder, similar in location to the benign FEP discovered previously (Fig. 1). Cystoscopy revealed a lobulated, polypoid lesion seemingly attached to the left bladder neck (Fig. 2). Transurethral resection of the mass was attempted but incompletely achieved due to anteriorly positioned mass. In addition to the left bladder neck, mass was contiguous with the left intraurethral prostatic lobe. Histologic examination of the entirely submitted specimen demonstrated a biphasic epithelial-stromal tumor with benign urothelial lining and malignant stromal cells displaying marked cytologic atypia, numerous mitoses, and necrosis. There was also morphologic and immunophenotypic evidence of rhabdomyoblastic differentiation (e.g. myogenin, myo-D1, and desmin positive), sharply contrasting the findings of the previously resected lesion at this site (Fig. 3). Although both lesions had a biphasic polypoid appearance with benign urothelial lining, the stromal component of the current lesion was frankly malignant and consistent with the diagnosis of a Grade 3 (FNCLCC grading) rhabdomyosarcoma (Fig. 4). There was no evidence of sarcomatoid carcinoma and no PAX3/FOXO1 or PAX7/FOXO1 fusion transcript was detected by RT-DNA amplification.
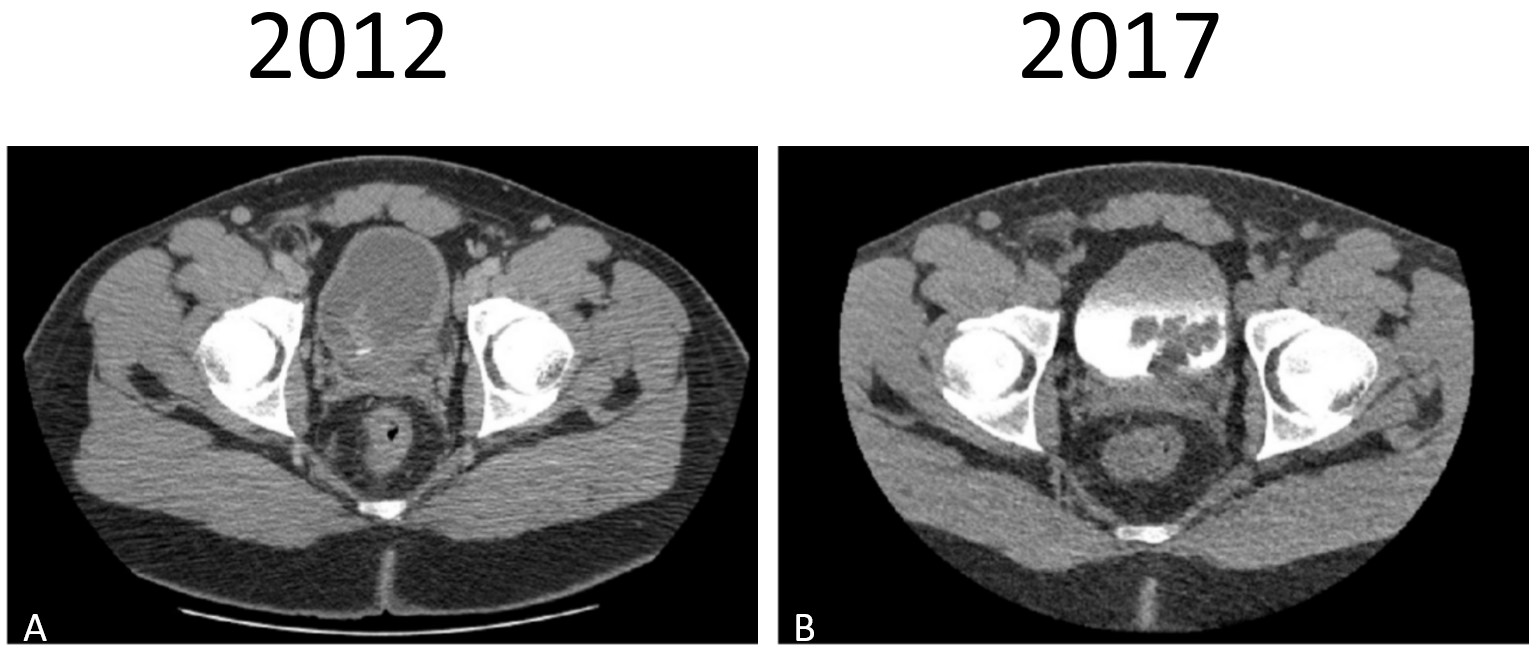
Figure 1: Computed tomography (CT) imaging. A) Fibroepithelial polyp (2012): Abdominal/pelvic CT with contrast demonstrating bladder wall thickening/mass lesion. B) Rhabdomyosarcoma (2017): Abdominal/pelvic CT with contrast demonstrating a 5x4cm filling defect in bladder.
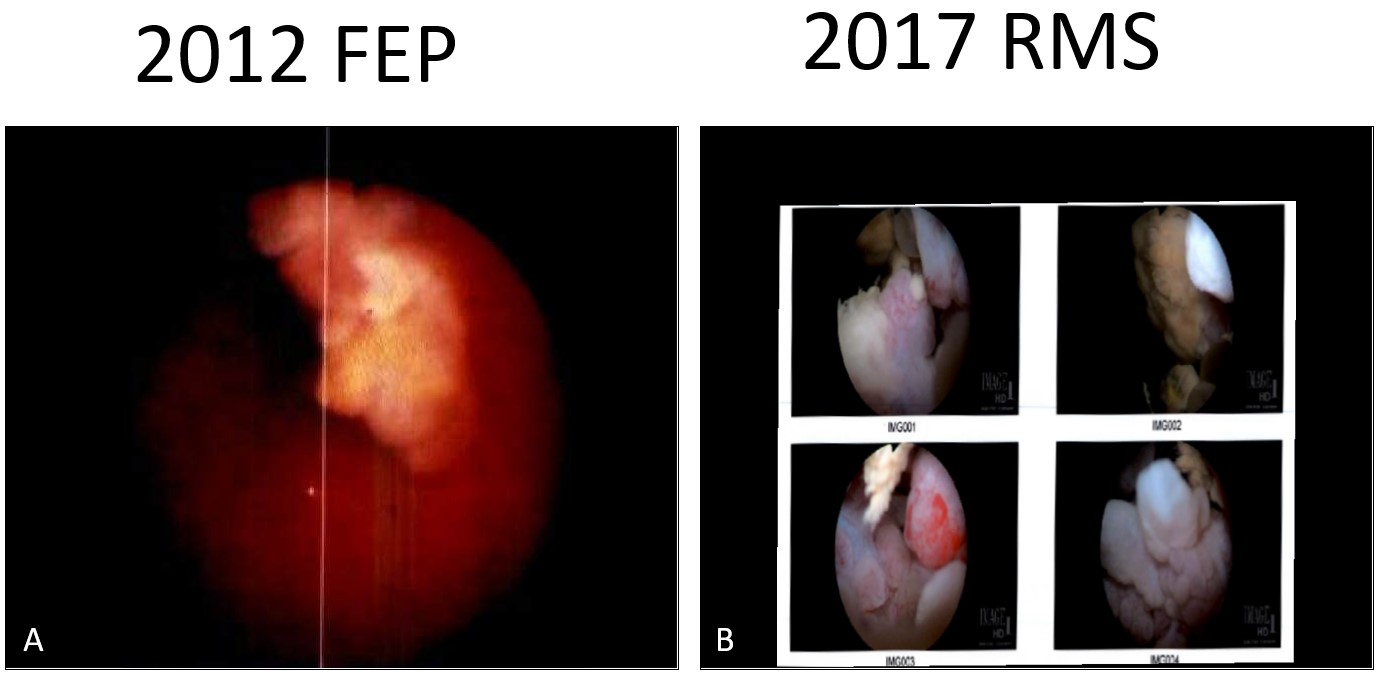
Figure 2: Cystoscopic imaging. A) Fibroepithelial polyp (2012): large lobulated mass on the posterior aspect of the prostate, protruding into the bladder. B) Rhabdomyosarcoma (2017): large polypoid structure connected to the left side of the prostate, emanating from the bladder neck.
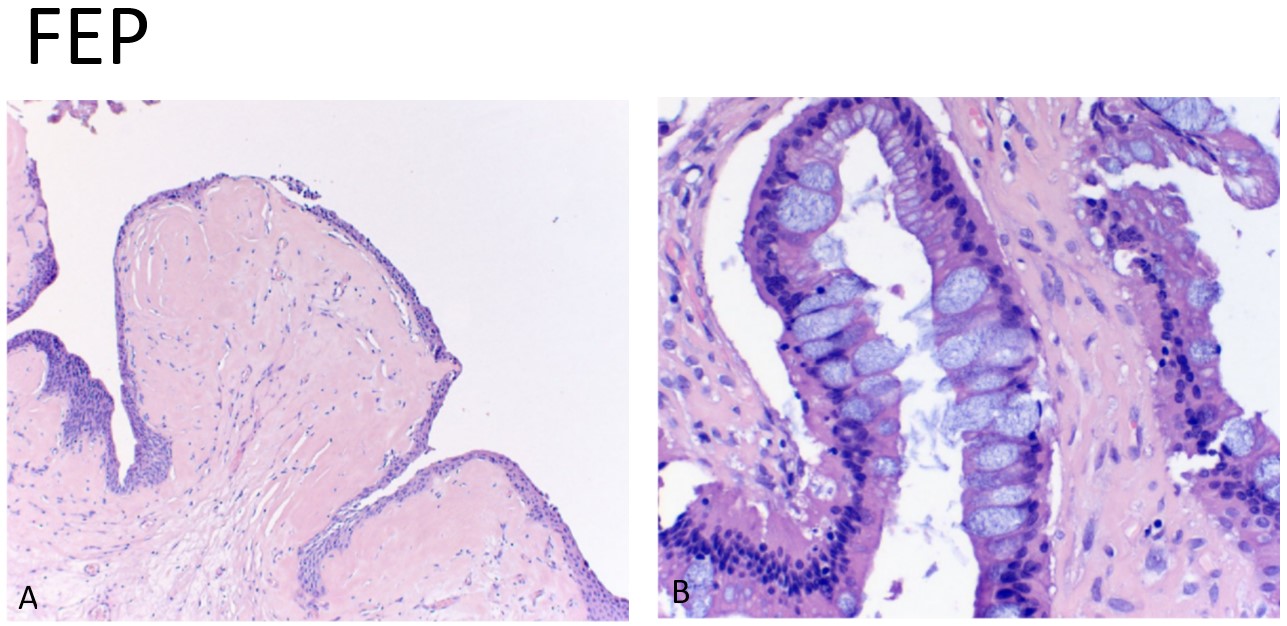
Figure 3: Hematoxylin and eosin (H&E) staining of the fibroepithelial polyp (2012): A) A polypoid tumor lined by benign urothelium and underlying hyalinized and acellular stroma. B) Extensive intestinal metaplasia without dysplasia also present.
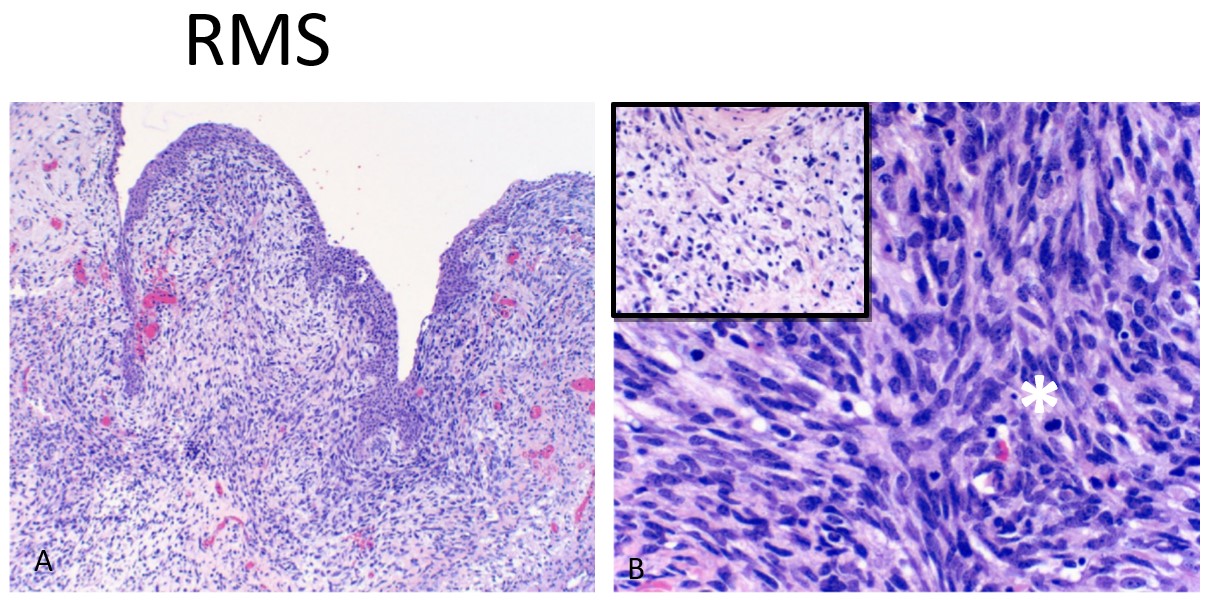
Figure 4: Hematoxylin and eosin (H&E) staining of the rhabdomyosarcoma (2017): A) A polypoid tumor lined by benign urothelium and underlying malignant stromal cells, B) with spindle and epithelioid morphology and numerous mitosis (*). Inset shows myxoid changes and rhabdomyoblastic differentiation with abundant eosinophilic cytoplasm.
Follow-up MRI of the pelvis demonstrated a 2.7x1.7 cm exophytic bladder mass with diffuse bladder wall thickening. Abnormal enhancement of the prostatic urethra and surrounding central prostate without discrete margins was also observed. No distant metastatic disease or lymphadenopathy was noted. After case discussion at multi-disciplinary tumor board and thorough patient counseling, the decision was made to proceed with exenterative surgery.
A robotic radical cystoprostatectomy with bilateral extended pelvic lymph node dissection and creation of orthotopic Studer ileal neobladder was performed under general anesthesia, ultimately with good patient recovery. Final pathology demonstrated rhabdomyosarcoma larger than 5.0 cm in greatest dimension with negative margins and no metastasis in 62 recovered lymph nodes (pT2bN0MxR0 based on AJCC 7th edition for soft tissue sarcomas). Given the absence of positive margins, lymph node involvement, or metastatic disease, there was no clear indication for patient to receive adjuvant chemotherapy or radiation therapy.
On follow-up with the Urology department at 4, 8, 12, and 24 months, CT abdomen/pelvis with and without contrast demonstrated no disease recurrence. His recent metabolic laboratory studies were unremarkable. Functionally, he was continent and potent.
Discussion
Rhabdomyosarcoma is a malignant soft tissue sarcoma, peaking between ages 2-6 and 10-18 (Dasgupta et al., 2016). The genitourinary tract only accounts for 15-20% of all RMS tumors (Ahsaini et al., 2018). Multimodal therapy using neoadjuvant chemotherapy, radiotherapy, and surgical consolidation has led to >70% 5-year overall survival (Dasgupta et al., 2016). To date, there has been limited research into the management of adults diagnosed with bladder RMS, owing to the extreme rarity of the disease. In fact, less than 50 patients are identified in medical literature.
The most common presenting symptoms of bladder RMS are hematuria, dysuria, bladder dysfunction, or symptoms resulting from mass effect (Ahsaini et al., 2018). In adults, the few case reports published demonstrated an array of outcomes ranging from improved disease free-survival after neoadjuvant chemotherapy and surgery or radiotherapy and surgery, to death. Poor outcomes usually resulted from subsequent disease recurrence after surgery, or other medical complications.
Histologically, RMS cells generally have high nuclear to cytoplasmic ratio with frequent mitotic figures and variable amount of necrosis. The cells may appear as primitive, uniform round blue cells, be pleomorphic and diffusely anaplastic, or show a predominantly spindle cell morphology. Architecturally, they may be arranged in sheets, alveoli, or large and small nests with associated myxoid matrix or collagenous/hyalinized matrix.
Conversely, benign FEP generally displays a biphasic urothelial/stromal morphology. In adults they are rare and may be misdiagnosed as a variety of more common reactive and neoplastic lesions. Three distinct histologic patterns have been described: as a polypoid mass with club-like projections and florid cystitis cystica et glandularis, numerous small papillae with densely fibrotic stroma, and polypoid lesion with tall fingerlike projections. No cytologic atypia is seen in the urothelial or stromal component, with the exception of stromal degenerative atypia. The biphasic nature of these lesions is reminiscent of phyllodes tumor, a well-recognized entity in female breast, that has been described in the male urogenital tract including prostate and seminal vesicle. A recent case report describes a phyllodes tumor of the urinary bladder in an adult female with longstanding history of recurrent hematuria and multiple resections of polypoid masses (Tchrakian et al., 2018). More recently, a DICER-1 mutation positive giant botryoid FEP of the urinary bladder has been described in an adult female exhibiting histologic resemblance to embryonal RMS with a botryoid appearance and prominent myxoid stroma (Eckstein et al., 2019). Interestingly, a case report from 1982 details an adult male with an FEP displaying predominately myxomatous appearance on initial biopsy, but eventually revealing rhabdomyoblasts and cross striations two-and-a-half years later after multiple additional resections (Dupree and Fisher, 1982). They suggested a histologic continuum between FEP and RMS, but acknowledge malignant transformation could not be ruled out. Currently, this is the only available follow-up data in the reported cases of the urinary bladder FEPs or phyllodes tumor that may suggest a possible progression or malignant transformation.
In the context of our patient, the initial resection of the polypoid tumor (soft tissue aggregating to 4.0 x 2.5 x 0.8cm, entirely submitted) had characteristic features of a benign FEP with benign urothelial lining and predominantly hyalinized/acellular stromal component. The patient had no known history of resection or previous injury, there was no inflammatory cell infiltration, and the associated extensive intestinal metaplasia showed no evidence of dysplasia or adenocarcinoma. Five years later, he presented with a large polypoid tumor in the same location. The subsequent resection specimen showed a biphasic lesion with a frankly malignant stromal component with high cellularity, round blue cells with occasional rhabdomyoblastic-appearing cells, numerous mitoses and necrosis. Extensive additional work-up supported the diagnosis of rhabdomyosarcoma, most compatible with the embryonal subtype. This case is unique in that, unlike previous reported adult bladder RMS cases that represent de novo malignancies (Rahoui et al., 2022; Knowles et al., 2022), it presents a temporally and anatomically correlated FEP with subsequent development of RMS. A SEER database study demonstrated that younger age and localized disease are associated with improved survival in adult RMS (Patel et al., 2020), while another study reported that the pleomorphic subtype is associated with a worse prognosis (Liu et al., 2019). Our patient, a relatively young 35 years old with localized, embryonal subtype disease, possesses several protective factors associated with improved outcomes. The benign polypoid lesion was found to be negative for DICER1 gene mutation, ruling out the possibility of a giant botryoid FEP. The intriguing presentation of this sequence of such rare lesions in the urinary bladder of adults (FEP and RMS) raises the possibility of malignant transformation.
Embryonal RMS is thought to arise de novo from dysregulation of primitive mesenchymal progenitor cells with skeletal myogenic differentiation pathways. These include RAS signaling pathway disruption (Martinelli et al., 2009) and overexpression of IGF2 at chromosome 11p15 (Venkatramani et al., 2016). There is a lack of molecular evidence pointing that RMS arises from pre-existing benign lesions, and no study has demonstrated a direct causal relationship between RMS FEP, despite isolated case reports describing temporal association with morphological evolution. Therefore, although our case demonstrates recurrence at the same anatomic site as over time, no causal inference can be made regarding malignant transformations from benign lesion. Instead, this case should be considered as hypothesis-generating, warranting further investigation into potential underlying tumorigenesis mechanisms.
While multimodal therapy seems to be the favored regimen in younger patients, there have not been enough studies in adult bladder RMS to evaluate one standardized approach. In our case, disease was localized to the bladder, allowing us to perform surgical resection with negative margins. The patient’s desire to maintain functional voiding per urethra via an orthotopic neobladder and the inopportune location of the tumor at the bladder neck—with risk of continued progression and forfeiture of the option of continent diversion—influenced the decision for immediate surgery rather than neoadjuvant chemotherapy with radiation. Thus far, the patient has had no evidence of disease recurrence with close to 3 years of follow-up. Given the lack of lack of reliable urine-based biomarker, long-term follow-up via bladder ultrasound and/or cystoscopy is warranted for early detection of recurrence.
Conclusion
This case highlights a rare presentation of adult bladder RMS and raises the question of possible malignant transformation from a prior benign FEP. The unusual temporal and spatial sequence add to the limited literatures suggesting morphological evolution, although a causal relationship remains unproven. Given the rarity of these tumors, standardized treatments are lacking. In light of the overlapping features presented in this case, it may be wise to consider long-term follow-up in patients presenting with a benign FEP, until further data can delineate the relationship between the benign FEP and malignant RMS.
Acknowledgments: No funding sources or other forms of assistance were necessary for the conduct of the study.
Author Disclosure Statement: We have no conflicts of interest or financial disclosures
Ethics Statement: Ethical review and approval were not required for the study on human participants in accordance with the local legislation and institutional requirements. Written informed consent was obtained from the patient for the publication of any potentially identifiable images or data included in this article.
References:
Ahsaini M, Ouattar K, Azelmad H, Mellas S, Ammari JE, Tazi MF, Fassi MJ, Farih MH, Sekal S, Harmouch T. A rare pure embryonal rhabdomyosarcoma of the urinary bladder in an adult successfully managed with neoadjuvant chemotherapy and surgery: a case report. J Med Case Rep 2018; 12: 329.
Dasgupta R, Fuchs J, Rodeberg D. Rhabdomyosarcoma. Semin Pediatr Surg 2016; 25: 276-283.
Dupree WB, Fisher C. Rhabdomyosarcoma of prostate in adult. Long-term survival and problem of histologic diagnosis. Urology 1982; 19: 80-82.
Eckstein M, Agaimy A, Woenckhaus J, Winter A, Bittmann I, Janzen J, Bertz S, Haller F, Hartmann A. DICER1 mutation-positive giant botryoid fibroepithelial polyp of the urinary bladder mimicking embryonal rhabdomyosarcoma. Hum Pathol 2019; 84: 1-7.
Hawkins WG, Hoos A, Antonescu CR, Urist MJ, Leung DH, Gold JS, Woodruff JM, Lewis JJ, Brennan MF. Clinicopathologic analysis of patients with adult rhabdomyosarcoma. Cancer 2001; 91: 794-803.
Knowles KJ, Avalos SR, Shi M, Parwani A, Holloway AR, Keel CE. Rhabdomyosarcoma of the urinary bladder in an adult: Case report and review of the literature. Urol Case Rep 2022; 45: 102169.
Liu YT, Wang CW, Hong RL, Kuo SH. Prognostic Factors and Treatment Outcomes of Adult Patients With Rhabdomyosarcoma After Multimodality Treatment. Anticancer Res 2019; 39: 1355-1364.
Martinelli S, McDowell HP, Vigne SD, Kokai G, Uccini S, Tartaglia M, Dominici C. RAS signaling dysregulation in human embryonal Rhabdomyosarcoma. Genes Chromosomes Cancer 2009; 48: 975-982.
Patel SR, Hensel CP, He J, Alcalá NE, Kearns JT, Gaston KE, Clark PE, Riggs SB. Epidemiology and survival outcome of adult kidney, bladder, and prostate rhabdomyosarcoma: A SEER database analysis. Rare Tumors 2020; 12: 2036361320977401.
Rahoui M, Ouanes Y, Marrak M, Chaker K, Ben Rhouma S, Nouira Y. Adult bladder rhabdomyosarcoma: A case report. Urol Case Rep 2022; 44: 102134.
Sultan I, Qaddoumi I, Yaser S, Rodriguez-Galindo C, Ferrari A. Comparing adult and pediatric rhabdomyosarcoma in the surveillance, epidemiology and end results program, 1973 to 2005: an analysis of 2,600 patients. J Clin Oncol 2009; 27: 3391-3397.
Tchrakian N, Browne E, Shanks JH, Flynn RJ, Crowther S. Phyllodes tumour of the urinary bladder: a report of a unique case. Histopathology 2018; 72: 356-358.
Venkatramani R, Triche TJ, Wang L, Shimada H, Mascarenhas L. Insulin-like Growth Factor 2 Gene Expression Molecularly Differentiates Pleuropulmonary Blastoma and Embryonal Rhabdomyosarcoma. J Pediatr Hematol Oncol 2015; 37: e356-60.
|




