FRONTIERS IN MEDICAL CASE REPORTS - Volume 7; Issue 2, (Mar-Apr, 2026)
Pages: 01-09
Print Article
Download XML Download PDF
EUS-Guided Radiofrequency Ablation for Pancreatic Insulinoma: A Case Report
Author: Gabriela Mintegui, Zara Martínez
Category: Medical Case Reports
Abstract:
Endogenous hyperinsulinemic hypoglycemia remains a diagnostic challenge requiring precise biochemical and imaging evaluation to determine its etiology. Insulinoma, although rare, is the most common functional pancreatic neuroendocrine tumor. It arises from pancreatic beta cells and is characterized by inappropriate insulin secretion, leading to recurrent hypoglycemia.
We report the case of a 40-year-old woman with recurrent symptomatic hypoglycemia in whom Whipple’s triad was documented. Endoscopic ultrasound identified a 13 × 12 mm lesion in the pancreatic neck, consistent with insulinoma. Given the clinical and anatomical characteristics, endoscopic ultrasound-guided radiofrequency ablation was performed as a minimally invasive therapeutic alternative.
The patient had a favorable short-term outcome, with complete resolution of symptoms, no residual lesion on imaging at 3 months, and normal fasting glucose, insulin, and C-peptide levels at 4 months.
Keywords: Hypoglycemia, Insulinoma, Radiofrequency Ablation
Full Text:
Introduction
Hypoglycemia in non-diabetic individuals (plasma glucose typically <55 mg/dL) represents a diagnostic challenge and requires a systematic clinical, biochemical, and etiological evaluation.
The presence of Whipple’s triad supports the diagnosis of pathological hypoglycemia and helps distinguish it from physiological decreases in blood glucose levels. This triad includes symptoms consistent with hypoglycemia, documented low plasma glucose during the episode, and resolution of symptoms after restoration of glucose to baseline levels (Hofland et al., 2024).
Hypoglycemic symptoms are classified into autonomic (adrenergic) and neuroglycopenic manifestations. Adrenergic symptoms, secondary to activation of the sympathetic nervous system and catecholamine release, include diaphoresis, tremor, palpitations, anxiety, nervousness, and increased appetite (Shin et al., 2010).
These symptoms typically occur when glucose levels fall below physiological counterregulatory thresholds, generally around 55 mg/dL. In contrast, neuroglycopenic symptoms result from inadequate glucose availability to the central nervous system, usually appearing at glucose levels below 45–50 mg/dL (Cryer, 2007), and include altered consciousness, cognitive impairment, transient focal neurological deficits, seizures, and coma (Shin et al., 2010).
Overall, these manifestations may occur during fasting, exercise, or even in the postprandial period, and in severe cases may be life-threatening (Crinò et al., 2023; Mintegui et al., 2026).
The counterregulatory response, including glucagon and epinephrine release, may be activated at plasma glucose levels approximately 10 mg/dL higher than those at which hypoglycemic symptoms appear (Cryer, 2007). Moreover, glycemic thresholds for these responses may be shifted to higher values in patients with insulinoma (Shin et al., 2010).
Among the causes of endogenous hyperinsulinemic hypoglycemia, insulinoma is the most common functional pancreatic neuroendocrine tumor, although it accounts for only 1–2% of all pancreatic neoplasms (Shin et al., 2010). Its incidence is estimated at 1–4 cases per million individuals per year, with a peak between the third and sixth decades of life and a slight female predominance (Hofland et al., 2024; Shin et al., 2010; Debraine et al., 2024; de Herder et al., 2006).
In most cases, insulinomas are sporadic, solitary, and benign tumors; however, approximately 10% are associated with hereditary syndromes, particularly multiple endocrine neoplasia type 1 (MEN1) (Shin et al., 2010). They originate predominantly from pancreatic islet beta cells and are characterized by inappropriate and autonomous insulin secretion.
The diagnosis of insulinoma remains challenging and is often delayed due to the nonspecific nature of its clinical manifestations and the small size of these lesions, requiring integration of biochemical and imaging studies.
Biochemical evaluation must demonstrate hypoglycemia accompanied by inappropriately elevated endogenous insulin levels relative to plasma glucose. A supervised fasting test of up to 72 hours remains the diagnostic gold standard (Okabayashi et al., 2013).
During this procedure, once plasma glucose falls below 55 mg/dL, insulin, C-peptide, proinsulin, and β-hydroxybutyrate should be measured simultaneously. Biochemical criteria consistent with insulinoma include insulin ≥6 μU/mL, C-peptide ≥2 ng/mL, and proinsulin ≥5 pmol/L (Placzkowski et al., 2009).
In suspected factitious hypoglycemia or insulin autoimmune syndrome, evaluation should include sulfonylurea screening and anti-insulin antibodies (Cryer, 2007).
Following biochemical confirmation, precise anatomical localization is essential. Conventional imaging modalities such as ultrasound, CT, and MRI may have limited sensitivity, particularly for tumors smaller than 2 cm (Placzkowski et al., 2009; Okabayashi et al., 2013).
When these are inconclusive, more sensitive techniques such as endoscopic ultrasound may be required.
Surgery remains the treatment of choice for localized insulinoma, with high cure rates (Shin et al., 2010; Masciangelo et al., 2021). However, in selected patients, endoscopic ultrasound-guided radiofrequency ablation has emerged as a minimally invasive therapeutic alternative with promising results.
Case Report
A 40-year-old woman with no relevant past medical history presented with recurrent episodes of dizziness, diaphoresis, and presyncope. She had been evaluated on multiple occasions in emergency departments without conclusive findings. During the course of her illness, she experienced a new episode while driving, with a transient loss of consciousness resulting in a traffic accident. She denied alcohol consumption, medication use, or exposure to hypoglycemic agents. She was referred to our center for a second opinion, as she had experienced recurrent episodes of hypoglycemia for three months, significantly limiting her daily activities and preventing her from leaving her home.
During one evaluation, Whipple’s triad was documented, with a plasma glucose level of 42 mg/dL and suppressed ketonemia. A supervised fasting test was performed but had to be interrupted early, at 4 hours, due to the onset of symptoms consistent with hypoglycemia. At that time, laboratory results showed a plasma glucose level of 48 mg/dL, with inappropriately elevated insulin (8.1 μU/mL) and C-peptide (2.91 ng/mL) levels, and a cortisol level of 16 μg/dL. Based on these findings, a diagnosis of endogenous hyperinsulinemic hypoglycemia was established, with suspicion of insulinoma.
For tumor localization, computed tomography (CT), CT angiography, abdominal magnetic resonance imaging, and positron emission tomography (PET) with somatostatin analogs were performed, all of which were inconclusive. Subsequently, ^18F-DOPA PET revealed a focal area of increased uptake in the pancreatic head region. Endoscopic ultrasound (EUS) provided precise anatomical characterization, demonstrating a hypoechoic, round, well-defined lesion located in the pancreatic neck, measuring 13 × 12 mm (Fig. 1). EUS-guided fine-needle aspiration was performed, and cytological analysis revealed features consistent with a well-differentiated neuroendocrine tumor. Immunohistochemical evaluation showed a low proliferative index (Ki-67 <3%), consistent with a grade 1 tumor (Fig. 2).
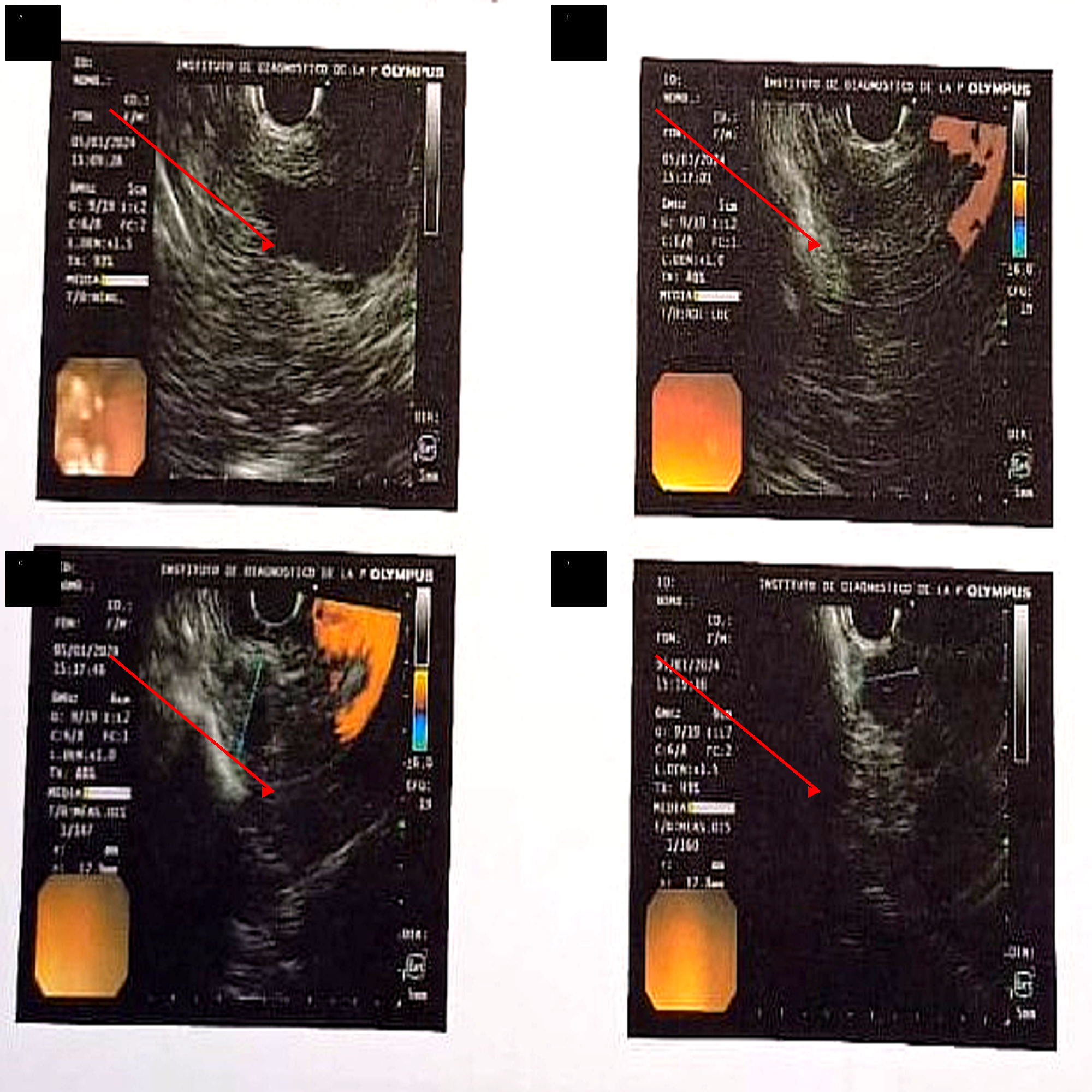
Figure 1: Endoscopic ultrasound (EUS) findings of pancreatic insulinoma located at the pancreatic neck.
(A–D) Hypoechoic, well-defined lesion (arrows) in the pancreatic neck, with internal vascularization on Doppler imaging and homogeneous echotexture.
EUS allowed precise localization and characterization of the lesion.
Figure 2: Endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) cytology of pancreatic insulinoma.
(A–D) Cytological specimens reveal cohesive clusters of monotonous, small-to-medium-sized cells with round nuclei, finely granular chromatin (“salt-and-pepper” pattern), and scant cytoplasm, consistent with a neuroendocrine neoplasm. Immunocytochemical staining shows diffuse positivity for CD56, supporting neuroendocrine differentiation. These findings are compatible with a well-differentiated pancreatic neuroendocrine tumor (insulinoma).
Tumor markers, including carcinoembryonic antigen (CEA), carbohydrate antigen 19-9 (CA 19-9), and chromogranin A, were within normal ranges. In the appropriate clinical context, these findings supported the diagnosis of insulinoma.
Following multidisciplinary evaluation, and considering tumor size, its location in the pancreatic neck, and the need to preserve pancreatic parenchyma, endoscopic ultrasound-guided radiofrequency ablation was selected.
Radiofrequency ablation was performed (6 weeks after the initial EUS). During the ablation procedure, the lesion measured 19.4 mm. The apparent increase compared with the initial EUS measurement (13 × 12 mm) should be interpreted cautiously, as it may reflect differences in measurement planes, probe positioning, and intraprocedural imaging conditions; however, interval growth over the 6-week period cannot be completely excluded.
At 3-month clinical and imaging follow-up, the patient remained asymptomatic, with no recurrence of hypoglycemic episodes, normal glucose levels, and no residual lesion on imaging studies.
At 4 months, biochemical reassessment demonstrated normal fasting glucose, insulin, and C-peptide levels, further supporting a sustained clinical and metabolic response. Ongoing follow-up has been planned, including clinical, biochemical, and imaging reassessment at 12 months, in line with current recommendations.
Although measurement variability is the most likely explanation, the possibility of interval growth cannot be excluded and highlights the importance of standardized imaging assessment.
At the time of submission, the patient remains under active follow-up without recurrence of symptoms.
Discussion
In individuals without underlying medical conditions, not receiving pharmacological treatment, and appearing in good general health, the main causes of hypoglycemia to consider include endogenous hyperinsulinemia and factitious hypoglycemia (Cryer, 2007; Cryer, 2010). Although uncommon, insulinoma represents the most relevant cause of endogenous hyperinsulinemic hypoglycemia and the most frequent functional pancreatic neuroendocrine tumor (Shin et al., 2010).
Its clinical significance lies not only in its low incidence but also in the potential severity of hypoglycemic episodes and the diagnostic difficulty resulting from frequently nonspecific clinical manifestations. These may be misinterpreted as neurological, psychiatric, or cardiovascular disorders, leading to diagnostic delays with potentially serious consequences (Shin et al., 2010). In the present case, recurrent hypoglycemic episodes significantly impaired the patient’s quality of life and culminated in a traffic accident secondary to transient loss of consciousness, highlighting the life-threatening risk associated with delayed diagnosis (Cryer, 2007).
Whipple’s triad remains a fundamental cornerstone in the diagnostic suspicion of insulinoma. In our patient, its documentation, together with suppressed ketonemia and biochemical confirmation of endogenous hyperinsulinism during the supervised fasting test, allowed diagnosis without the need to complete a prolonged 72-hour fast. This has important clinical implications, as it avoids unnecessarily prolonging a potentially uncomfortable and not risk-free procedure in patients with early onset of symptoms. This approach is consistent with the literature (Shin et al., 2010; Cryer, 2007; Cryer, 2010).
Transabdominal ultrasound and computed tomography (CT) are commonly used initial imaging modalities in the evaluation of insulinoma. However, their sensitivity may be limited, particularly in tumors smaller than 2 cm (Placzkowski et al., 2009; Okabayashi et al., 2013).
More sensitive methods should be considered when conventional imaging fails to localize the lesion. Endoscopic ultrasound is a highly sensitive and specific technique for detecting pancreatic neuroendocrine tumors and is recommended once the clinical and biochemical diagnosis has been established. These findings are consistent with our case, in which endoscopic ultrasound enabled precise identification of the lesion after negative results from conventional imaging studies.
Other imaging modalities include magnetic resonance imaging and positron emission tomography with ^18F-DOPA, which may be particularly useful in lesions not detected by conventional studies. Additionally, Ga-68 DOTATATE PET/CT can be used in selected cases, although its performance may be limited in some insulinomas.
Tumor localization also represented a diagnostic challenge in our case. CT, CT angiography, abdominal magnetic resonance imaging, and PET with somatostatin analogs failed to identify the lesion, whereas ^18F-DOPA PET suggested localization and endoscopic ultrasound provided precise anatomical definition and allowed cytological confirmation.
Surgery remains the standard treatment for localized insulinoma, with cure rates exceeding 90% (Shin et al., 2010; Masciangelo et al., 2021). However, pancreatic surgery is not free of morbidity, particularly in anatomically complex locations such as the pancreatic neck.
In this context, endoscopic ultrasound-guided radiofrequency ablation has emerged as a minimally invasive therapeutic alternative in selected patients, particularly in small, well-defined lesions. Its main advantage lies in achieving tumor control while preserving pancreatic parenchyma and reducing surgical morbidity.
In our patient, early biochemical reassessment at 4 months confirmed normalization of fasting glucose, insulin, and C-peptide levels, reinforcing the clinical and imaging findings and supporting the short-term effectiveness of the procedure.
However, longer follow-up is required to confirm sustained remission, as recurrence or residual disease may not be evident in the early post-procedural period. Current recommendations suggest clinical, biochemical, and imaging reassessment for at least 12 months after ablation.
This case contributes to the growing evidence supporting EUS-guided radiofrequency ablation as a viable alternative in selected patients, particularly when surgery poses increased risk or when organ preservation is prioritized.
Conclusions
This case highlights the importance of considering insulinoma in the differential diagnosis of recurrent hypoglycemia and underscores the value of integrated clinical and imaging evaluation.
While surgery remains the standard treatment, endoscopic ultrasound-guided radiofrequency ablation may represent a promising and minimally invasive alternative in carefully selected patients.
Ethics Statement: Written informed consent was obtained from the patient for publication of this case report and any accompanying images, according to institutional policy. The study was conducted in accordance with the ethical principles of the Declaration of Helsinki and applicable international standards for case report publication.
Conflict of Interest: The authors declare that there are no conflicts of interest.
Data Availability Statement: The data supporting the findings of this study are available from the corresponding author upon reasonable request.
References:
Crinò SF, Napoleon B, Facciorusso A, Lakhtakia S, Borbath I, Caillol F, et al. Endoscopic ultrasound-guided radiofrequency ablation versus surgical resection for treatment of pancreatic insulinoma. Clin Gastroenterol Hepatol 2023; 21: 2834–2843.e2.
Cryer PE. Hypoglycemia in diabetes. In: Holt RIG, Cockram C, Flyvbjerg A, Goldstein BJ, editors. Textbook of Diabetes. 4th ed. Oxford: Blackwell Publishing; 2010. p. 528–548.
Cryer PE. Hypoglycemia, functional brain failure, and brain death. J Clin Invest 2007; 117: 868–870.
de Herder WW, Niederle B, Scoazec JY, Pauwels S, Kloppel G, Falconi M, Kwekkeboom DJ, Oberg K, Eriksson B, Wiedenmann B, Rindi G, O'Toole D, Ferone D; Frascati Consensus Conference; European Neuroendocrine Tumor Society. Well-differentiated pancreatic tumor/carcinoma: insulinoma. Neuroendocrinology 2006; 84: 183-188.
Debraine Z, Borbath I, Deprez P, Bosly F, Maiter D, Furnica RM. Long-term clinical and radiological outcomes of endoscopic ultrasound-guided radiofrequency ablation of benign insulinomas. Clin Endocrinol (Oxf) 2024; 101: 485–490.
Hofland J, Refardt JC, Feelders RA, Christ E, de Herder WW. Approach to the patient: insulinoma. J Clin Endocrinol Metab 2024; 109: 1109–1118.
Masciangelo G, Campana D, Ricci C, Andrini E, Rakichevikj E, Fusaroli P, Lisotti A. Endoscopic Ultrasound-Guided Locoregional Treatments for Pancreatic Neuroendocrine Neoplasms. Curr Oncol 2025; 32: 113.
Mintegui G, Cabral S, Casella C. Malignant insulinoma: case report and literature review. Medi Clin Case Rep J 2026; 1: 1564-1568.
Okabayashi T, Shima Y, Sumiyoshi T, Kozuki A, Ito S, Ogawa Y, Kobayashi M, Hanazaki K. Diagnosis and management of insulinoma. World J Gastroenterol 2013; 19: 829-837.
Placzkowski KA, Vella A, Thompson GB, Grant CS, Reading CC, Charboneau JW, Andrews JC, Lloyd RV, Service FJ. Secular trends in the presentation and management of functioning insulinoma at the Mayo Clinic, 1987-2007. J Clin Endocrinol Metab 2009; 94: 1069-1073.
Shin JJ, Gorden P, Libutti SK. Insulinoma: pathophysiology, localization and management. Future Oncol 2010; 6: 229–237.
|


